If you’ve ever had your bone density tested, you’ve heard of a DXA scan. For years it’s been the “gold standard” for measuring bone density and diagnosing or assessing your risk of osteoporosis, sometimes in conjunction with a clinical risk questionnaire.
But what if I told you it’s not showing you the full picture of your bone health. As the Head of the Research and Development of the Interdisciplinary Center of Bone Diseases at Lausanne University Hospital in Switzerland, and the principal investigator of an ongoing cohort referred to as the OsteoLaus Study, I’m shedding light on how combining older and newer bone assessment tools can help better detect osteoporosis and assess fracture risk earlier.
In fact, there’s one test you should have done immediately after your DXA scan — it can even be done on the same machine using the same images (more on that later).
Although the OsteoLaus Study is ongoing, I’m excited to share my findings with you so you can stay on top of your bone health!
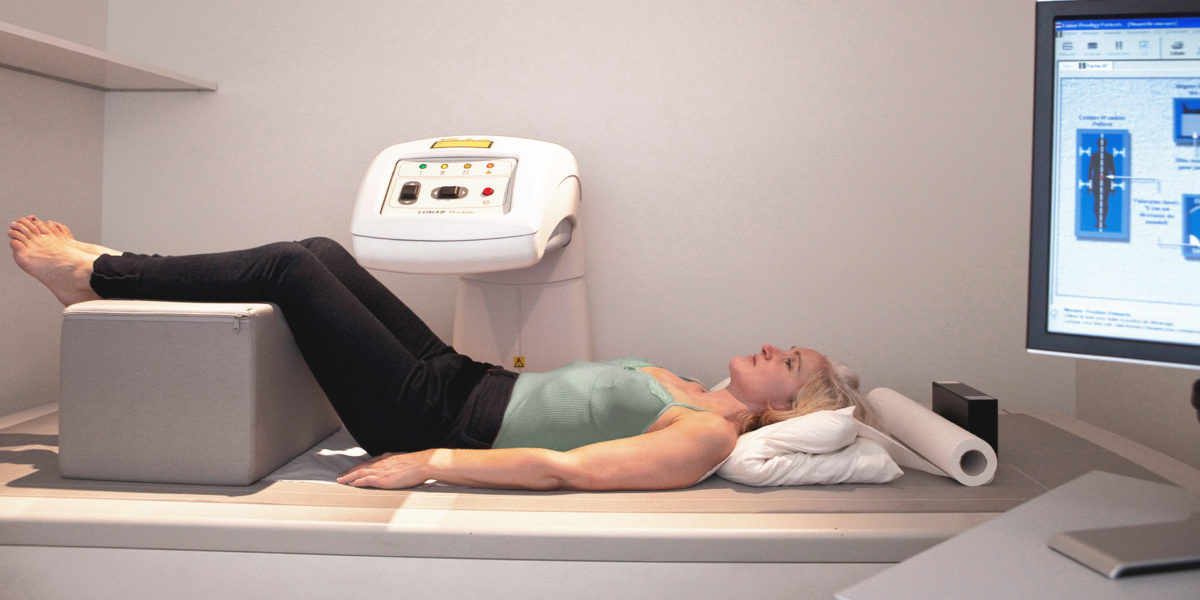
The OsteoLaus Study
The OsteoLaus Study is an ongoing prospective nested population-based cohort of 1,475 postmenopausal women (mean age of 64.4 ± 7.5 years) living in the city of Lausanne, Switzerland.1
The study is conducted by the Interdisciplinary Centre of Bone Diseases in close collaboration with the University of Lausanne. It’s designed to study bone health and aims to contribute to the early and accurate detection of increased fracture risk. In doing so, it seeks to contribute to the development of strategies for optimizing bone health care in postmenopausal women using classical and Artificial intelligence (AI) approaches.
Why the Study is Critical to the Earlier Detection of Bone Loss
Osteoporosis and fracture risk are not just fatalities related to aging and menopause. It is important to realize that osteoporosis and potential related fractures are preventable and treatable. To detect fracture risk and diagnose osteoporosis early and accurately, you need to have the proper tools.
Currently, DXA scans are widely used to measure bone mineral density (BMD) for osteoporosis diagnosis and fragility fracture risk prediction. But BMD only assesses the quantity of bone. So its relevance in the estimation of bone strength is limited.2
A significant concern with the use of DXA images is that they’re two-dimensional (2D) projections of the three-dimensional (3D) bone structure. And it’s a mathematical challenge to translate the 2D image of bone into the 3D structure that it represents.

So the trabecular bone score (TBS) was developed one decade ago to address both the challenging estimation of the actual 3D bone structure properties from the 2D DXA images and the limitations of BMD in osteoporosis diagnosis and fracture risk prediction.3
Studies have robustly shown that TBS predicts osteoporotic fractures independently of BMD and clinical risk factors.4, 5 So to help us accurately assess the bone health of our participants we knew we had to include TBS in our study. But we didn’t stop there.
The OsteoLaus Study includes comprehensive bone and body composition assessments for potential fracture or osteoporosis risk factors and beyond, such as:
- Body composition (including fat and lean parameters and their distribution)
- Data on anthropometrics, physical activities, and nutrition
- Fracture risk assessment (FRAX)
- Hip structural analysis (HSA)
- Physical function
- Full body, lumbar spine, and hip dual X-ray absorptiometry (DXA)
- Vertebral fracture Assessment (VFA)
- Heel quantitative ultrasound
How the Study is Structured
The OsteoLaus study focuses on three primary areas of research:
- Osteoporosis diagnosis
- Bone health of postmenopausal women
- Fragility fracture risk estimation in the Swiss population (including clinical risk factors, bone health, and muscle-related parameters)
The study’s priorities include:
- The development and validation of new likely tools for osteoporosis management
- The utility of current bone assessment tools for fracture risk prediction and osteoporosis diagnosis
- The prevalence and incidence of osteoporosis and osteoporotic fractures in postmenopausal women
- The determination of the association of bone health outcomes with cardiovascular diseases and mental and sleep disorders
The OsteoLaus study consists of a baseline visit and is projected to have at least four follow-up visits. The mean follow-up period between visits is two and a half years with a planned total follow-up time of ten years.
During the baseline visit (March 2010-December 2012), social, genetic, and biological data were also collected for all OsteoLaus participants. At baseline and the follow-up visits (first follow-up: September 2012 to June 2015; second follow-up: March 2015 to February 2018; and third follow-up: January 2018–2020), each participant was required to complete several dedicated questionnaires and underwent all the exams mentioned above.
Additionally, the TBS, HSA, and the 10-year probability of fracture estimated using the FRAX were also calculated.
What the Study Reveals So Far
During this study, participants had their BMD and TBS calculated for each of the L1–L4 vertebrae. Then lumbar spine (LS) BMD and TBS were calculated again, excluding vertebrae. BMD measurements were recorded for the total body, hip, and the femoral neck (FN) vertebrae using DXA scans.
At baseline, of the 1,475 women, 458 fractures were reported, including 183 major osteoporotic fractures (MOFs) – mostly vertebral fractures.
The incident MOFs were assessed from vertebral fracture assessments. Of the 1,475 women participants, 132 women experienced an MOF. Among the fractures, there were seven hip, 15 humerus, 33 forearm, and 86 vertebral fractures.
From the 132 women, nine had two MOFs, from which four had one forearm and one vertebral fracture; two had one forearm and one humerus fracture; two had one hip and one vertebral fracture; and one woman had one humerus and one vertebral fracture; all others had only one MOF.
Those who fractured were older. They also had more visceral fat and thicker soft tissue. Additionally, they had lower BMD at the LS, FN, and hip; had lower TBS; and had higher FRAX values than those who did not fracture.6 7
Our findings in the OsteoLaus Study validate the importance of using current as well as newer bone assessment tools for osteoporosis diagnosis and for fracture risk prediction and osteoporosis diagnosis. As the study is still ongoing, I can’t wait to share more interesting findings during the next follow-up!
Some Recent Findings: The Link Between Sleep, Bone Health, and TBS
Study 1: The Impact of Sleep on Bone Health
So from our OsteoLaus Study, we know that TBS is a great tool to help us accurately identify osteoporosis and associated fracture risk. Well, it turns out that TBS is also shedding light on another crucial aspect of our health — our sleep.
As you know, sleep is critical to your health. This biological process is involved in the regulation of a variety of endocrine and metabolic functions, including bone health. And previous studies have reported an association between sleep duration and quality and BMD in women.

In fact, a 2021 study investigated the relationship between sleep quality, sleep duration, and TBS in a general population of elderly adults. Among the 5,534 participants, 58.1% were women with a mean age of 66 years who had a sleep duration of 6.8 hours.
All participants underwent DXA assessment from which TBS from the lumbar spine was estimated. Also, their sleep quality was assessed using the Pittsburgh Sleep Quality Index. To test the association between TBS and all the sleep parameters, the researchers conducted linear regression analyses adjusted for many covariables including age, sex, cohort effect, BMI, education level, cholesterol levels, diabetes status, alcohol consumption, depression symptoms, smoking habits, and history of cardiovascular disease.
It was found that 28.5% of participants were poor sleepers. And a significant interaction was identified between depressive symptoms, sleep duration, and sleep quality. Moreover, participants classified as poor sleepers without depression symptoms had lower bone quality as assessed by TBS compared to good sleepers — but not observed in those with depressive symptoms.
Longer duration of sleeping was associated with higher TBS, but not among individuals with depressive symptoms. It was therefore concluded that poor sleep quality and duration are associated with low TBS (only in individuals without depression symptoms).8
Study 2: Insomnia’s Link to Osteoporosis
Another recent study showed that poor sleep is detrimental to several body systems, including bone.
In a 2-year prospective study, 190 Indian postmenopausal women with a mean age of 58.2 recorded their sleep duration and sleep quality using self-reporting and the Women’s Health Initiative Insomnia Rating Scale.
Anthropometry, bone biochemistry including bone turnover markers, BMD, and TBS were assessed at baseline and at the end of 2 years.
Among the participants, 34.2% had insomnia and 10.5% developed osteoporosis on follow-up.
The percentage decline over 2 years, in BMD of women with insomnia, was significantly higher at the lumbar spine (4.5 versus 1.6%) and femoral neck (2.9 versus 1.2%). The decrease in TBS was also significantly higher in women with suboptimal sleep (1.5 versus 0.5%) when compared to those with adequate sleep.
On multivariate analysis, insomnia was the sole factor that was predictive of osteoporosis. In conclusion, poor sleep quality was associated with osteoporosis in the participants.9 This finding suggests that getting optimal sleep may help to slow ongoing bone loss that results from sleep deprivation. However, given the small sample size further research is required.

Healthy bones start here
AlgaeCal provides the industry’s leading supplements and support designed to meet you at every stage of your bone-health journey. Find the right choice for you.
Takeaways
Although the OsteoLaus study is ongoing, it is already providing valuable insight into the role that older and newer bone assessment tools play in osteoporosis diagnosis and fracture risk prediction.
Although bone density measurements using DXA devices alone are still the common practice for osteoporosis diagnosis, the microarchitectural aspect of the bone is missing to fulfill the WHO definition of osteoporosis. Historically, we have not had a clinical tool to speak to bone microarchitecture. The OsteoLaus study confirms the importance of getting both a bone density measurement and TBS to stay on top of your bone health. The beauty of having both tests is that you get a better picture of your bone health: bone quantity and bone quality.
It’s been shown that the combination of TBS and bone density is better at predicting fractures than either test alone. So I wholeheartedly recommend you do both. It’s very easy to do so because you can get your TBS test done at the same time you get your DXA scan. It’s done on the same machine, using the same images, without extra ionizing radiation nor exam time for you.
With that being said, not all facilities offer TBS yet. But you can check here to see if a clinic near you does. You’ll also want to check with your health insurance company to see if it covers TBS, as CPT codes have existed since January 2022. If it doesn’t, the cost is about $50–$150 U.S. dollars.
And don’t overlook the importance of getting a good night’s sleep. As discussed earlier, your bone health is strongly linked to the quality and duration of your sleep and to your overall lifestyle too.
To learn more about healthy aging and bone health, sign up for our newsletter and receive weekly updates.
References
- Enisa Shevroja, Pedro Marques-Vidal, Bérengère Aubry-Rozier, Gabriel Hans, Fernando Rivadeneira, Olivier Lamy, Didier Hans “Cohort Profile: The OsteoLaus study,” International Journal of Epidemiology, Volume 48, Issue 4, August 2019, Pages 1046–1047g, https://doi.org/10.1093/ije/dyy276
- Marshall D, Johnell O, Wedel H. Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ. 1996;312(7041):1254–9. https://pubmed.ncbi.nlm.nih.gov/8634613/
- Hans D, Barthe N, Boutroy S, Pothuaud L, Winzenrieth R, Krieg MA. Correlations between trabecular bone score, measured using anteroposterior dual-energy X-ray absorptiometry acquisition, and 3-dimensional parameters of bone microarchitecture: an experimental study on human cadaver vertebrae. J Clin Densitom. 2011;14(3):302–12. https://pubmed.ncbi.nlm.nih.gov/21724435/
- Silva BC, Leslie WD, Resch H, et al. Trabecular bone score: a noninvasive analytical method based upon the DXA image. J Bone Miner Res. 2014;29(3):518–30. https://pubmed.ncbi.nlm.nih.gov/24443324/
- McCloskey EV, Oden A, Harvey NC, et al. A meta-analysis of trabecular bone score in fracture risk prediction and its relationship to FRAX. J Bone Miner Res. 2016;31(5):940–8. https://pubmed.ncbi.nlm.nih.gov/26498132/
- Enisa Shevroja,Bérengère Aubry-Rozier,Gabriel Hans,Elena Gonzalez-Rodriguez,Delphine Stoll,Olivier Lamy,Didier Hans, “Clinical Performance of the Updated Trabecular Bone Score (TBS) Algorithm, Which Accounts for the Soft Tissue Thickness: The OsteoLaus Study,” First published: 16 August 2019 https://asbmr.onlinelibrary.wiley.com/doi/10.1002/jbmr.3851
- https://academic.oup.com/ije/article/48/4/1046/5259223
- Artemis Gkitakouam Fjorda Koromania, Katerina Trajanoska, Enisa Shevrojac, M. Carola Zillikensa, Ling Oeiab, Annemarie Luik, Didier Hans, Fernando Rivadeneira, “Association between trabecular bone score and sleep patterns: Findings from a cohort study” Volume 14, Supplement, April 2021, 100828 https://www.sciencedirect.com/science/article/pii/S2352187221000838
- Kripa Elizabeth Cherian,Nitin Kapoor,Thomas Vizhalil Paul “Disrupted Sleep Architecture Is Associated With Incident Bone Loss in Indian Postmenopausal Women: A Prospective Study” First published: 26 July 2022 https://asbmr.onlinelibrary.wiley.com/doi/abs/10.1002/jbmr.4662






Letty Petrides
March 11, 2023 , 7:49 pmI live in perth. I would love to have a TBS, however on one dies it here as the doctors say the dexa was enough.